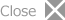Laboratory of organometallic and coordination chemistry
1 | Synthesis of linearly ordered multinuclear complexes designed to applications for nanoscale molecule devices
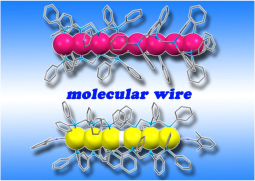
Extended metal atom chains (EMACSs) have attracted increasing attentions in relation to their unusual electronic, optical, and magnetic properties with respect to metal strings and potential applications for nanoscale molecule devices such as molecular wire, transistors, and sensors. However, their examples are extremely limited due to difficulties in their syntheses and characterization. We have successfully synthesized linear ordered tri-, tetra-, hexa-, and octanuclear complexes supported by methylene-bridged multidentate phosphine ligands. We have studied hexanualer platinum and octanuclear palladium complexes for the application of molecular wire. Furthemore, we have recently reported tetranuclear gold complexes are strongly luminescent and their strong emissions are sensitively quenched by chloride anions.
2 | Development of new reactions with multinuclear active centers
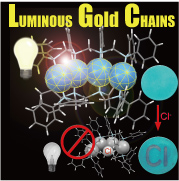
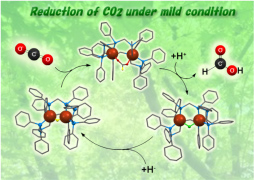
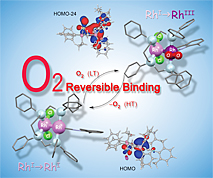
Transition metals have their own unique reactivities and are widely used as catalysts for mass production of medicine, agrichemicals, and electronic materials in the industrial chemistry. However, these catalysts are mononuclear complexes in many cases. In contrast, transition-metal clusters have attracted considerable interest due to their diverse properties such as catalyst, electronic, and magnetic devices induced by synergistic effects between adjacent metal centers, which are not established by mononuclear complexes. Our group has synthesized various homo- and heteromultinuclear structures supported with multidentate phosphine ligands, aiming to develop environmentally benign and energy saving catalytic process. We have recently found reduction of CO2 to formic acid under mild condition by a dinuclear copper complex and reversible dioxygen binding on dinuclear rhodium centers.
3 | Synthesis of multinuclear complexes aimed at enzyme mimic
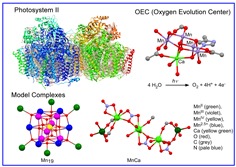
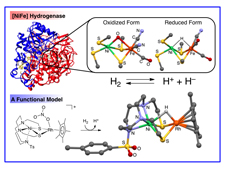
Enzymes are known to catalyze important metabolic processes very effectively and selectively which sustain life. Recently metal clusters have been found in the active centers of some enzymes by X-ray structure determinations. For examples, oxygen evolution center (OEC) in Photosystem II, where plants convert water to O2 under sun light, contain Mn4CaO5 core. [NiFe] Hydrogenases are able to catalyze heterolytic activation of H2 into a proton and hydride, and their active sites consist of Ni-Fe dimetal centers bridged by two thiolate ligands. Nirogenases have Fe7Mo clusters in active centers, which can reduce atomospheric nitrogen gas to ammonia under very mild condition. Our group has made an effort to synthesize model complexes similar to structures and reactivities of active sites with an aim to develop energy-efficient catalytic reaction processes.